

EnergyBatteries do not create energy, they store it. |
Batteries do not create energy, they store it. Think of a battery as a bucket that stores its contents. Just like a bucket, the battery needs to be filled to fulfil its role. A battery that is not regularly charged will stop working.
Batteries consist of cells. The battery case is most commonly made of plastic. Within the battery case is the cell packet, which consists of positive lead plates. There are also grills, separators and binders, as well as battery acid. It is the reaction between the battery acid and the lead plates that enables batteries to store electricity.
A fully charged cell has a voltage of 2.12V. This means that a fully charged, six-cell battery has a total voltage of 12.72V. A three-cell battery has a voltage of 6.36V.
If a 12V battery is "half-charged", it holds a charge of approximately 12.4V. If the battery is left at this voltage for for a significant period of time, it will become runied.
When a battery is discharged, the lead and lead oxide previously found in the lead plates transform into lead sulphate. This lead sulphate forms water, which dilutes the sulphuric acid. The accumulation of lead sulphate is an entirely normal part of a battery's process. When a battery is charged, this lead sulphate is turned back into lead, lead oxide and sulphuric acid.
If the battery is left uncharged for a long time, lead sulphate crystals will form to the extent that sulphation may occur. Sulphation can completely ruin a battery as it is impossible to transform such large crystals back into lead, lead oxide and sulphuric acid. When sulphation occurs, batteries rust.
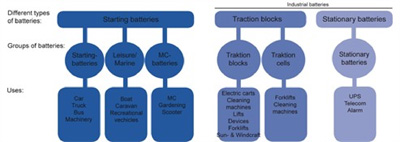
Batteries in our catalogue can be divided into three main types. Starting batteries, stationary batteries and traction batteries.
These have completely different characteristics, and are thus suitable for different purposes. Starting batteries are most common, as every car has one. Traction batteries and stationary batteries are not normally employed for private use and are therefore called industrial batteries.